Gold Nanoclusters in Cancer Photothermal Therapy: Mechanisms, Theranostic Applications, and Clinical Translation Over the Last Decade
DOI:
https://doi.org/10.65329/wjeb.v13.02.007Keywords:
Gold nanoclusters (AuNCs), Nanotheranostics, Photothermal conversion efficiency, Photothermal therapy, Precision oncology.Abstract
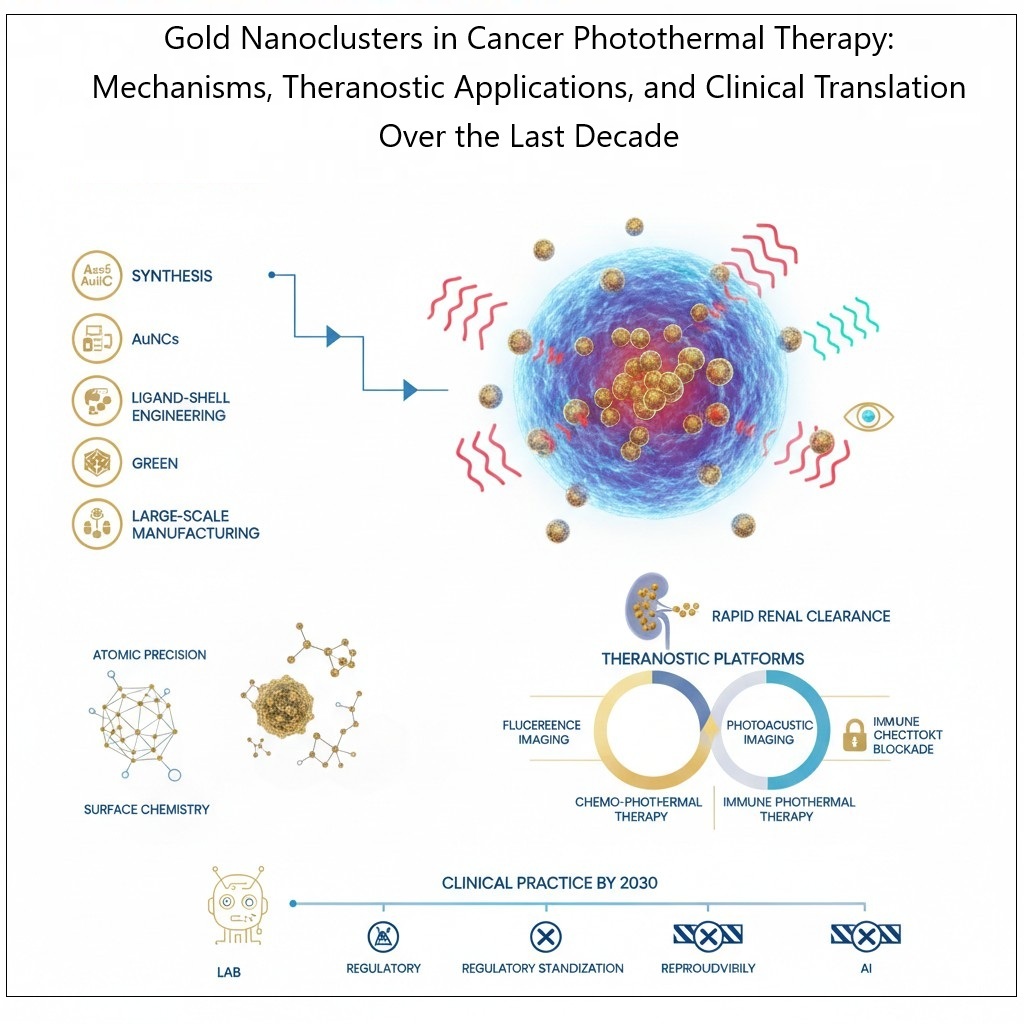
This last decade has seen the rise of gold nanoclusters (AuNCs) as a new, revolutionary class of photothermal agents for cancer therapy. The AuNCs (<2 nm) have a high surface area-to-volume ratio and exhibit efficient electron-to-phonon coupling. AuNCs possess favorable renal clearance properties due to their extremely small hydrodynamic diameters. This review article provides a structured summary of advancements made in the areas of AuNC synthesis, ligand-shell engineering, environmentally friendly fabrication methodologies, and large-scale manufacturing methods during the years of 2015-2025. It also focuses on the role atomic precision and surface chemistry play in the photothermal performance, biodistribution, protein corona formation, and nano–bio interactions of AuNCs; in addition, we highlight the mechanistic details associated with non-radiative relaxation pathways, energy gap modulation, and optimizing photothermal conversion efficiency. It also provides proof of concept that AuNCs are suitable as multifunctional theranostic platforms that integrate both fluorescence and photoacoustic imaging into combination strategies of chemo-photothermal and immune-photothermal therapy, with immune checkpoint blockade to elicit a systemic immune response against tumor cells. The study demonstrates rapid renal elimination and low accumulation of AuNCs over long-term duration, while addressing some of the historical concerns associated with traditional inorganic nanoparticles. Finally, the translational barriers facing AuNCs were highlighted, including regulatory standardization, reproducibility, and cost-effectiveness. The future with artificial intelligence (AI) will support AuNCs synthesis Thus, AuNC-based photothermal therapy is an exciting new advancement in nanomedicine and is predicted to be on a fast track for use in precision oncology from the lab through to integration into clinical practice by 2030.
References
[1] Yazbeck V, Alesi E, Myers J, Hackney MH, Cuttino L, Gewirtz DA. (2022) An overview of chemotoxicity and radiation toxicity in cancer therapy. Adv Cancer Res 155:1-27. https://doi.org/10.1016/bs.acr.2022.03.007. PMID: 35779872.
[2] Parambil BC, Moulik NR, Gollamudi VRM, Srinivasan S, Dhamne C, et al. (2025) Changing paradigms in pediatric cancer care - the contemporary landscape and perspectives for India. Ecancermedicalscience 19:1931. https://doi.org/10.3332/ecancer.2025.1931 .2025.1931. PMID: 40606948; PMCID: PMC12221250.
[3] Li M, Hasan I, Zhang L, Peng T, Guo B, Wang Z. (2025) Photothermal and Combinatory Therapy as an Emerging Therapeutic Paradigm of Breast Cancer Treatment. Int J Nanomedicine 20:13955-13988. https://doi.org/10.2147/IJN.S551712 . PMID: 41292550; PMCID: PMC12642939.
[4] Sharma N, Mohammad W, Le Guével X, Shanavas A. (2024) Gold Nanoclusters as High Resolution NIR-II Theranostic Agents. Chem Biomed Imaging 2(7):462-480. https://doi.org/10.1021/cbmi.4c00021. PMID: 39473532; PMCID: PMC11503699.
[5] Farkhani SM, Dehghankelishadi P, Refaat A, Gopal DV, Cifuentes-Rius A, Voelcker NH. (2024) Tailoring gold nanocluster properties for biomedical applications: From sensing to bioimaging and theranostics. Prog Mater Sci 142: 101229. https://doi.org/10.1016/j.pmatsci.2023.101229.
[6] van de Looij SM, Hebels ER, Viola M, Hembury M, Oliveira S, Vermonden T. (2022) Gold Nanoclusters: Imaging, Therapy, and Theranostic Roles in Biomedical Applications. Bioconjug Chem 33(1):4-23. https://doi.org/10.1021/acs.bioconjchem.1c00475. PMID: 34894666; PMCID: PMC8778645
[7] Loynachan CN, Soleimany AP, Dudani JS, Lin Y, Najer A, et al. (2019) Renal clearable catalytic gold nanoclusters for in vivo disease monitoring. Nat Nanotechnol 14(9):883-890. https://doi.org/10.1038/s41565-019-0527-6. PMID: 31477801; PMCID: PMC7045344..
[8] Zhou Y, Chen L, Wang T. (2025) Nanoparticles in gynecologic cancers: a bibliometric and visualization analysis. Front Oncol 14:1465987. https://doi.org/10.3389/fonc.2024.1465987. PMID: 39845315; PMCID: PMC11750695.
[9] Hao L, Dong C, Yu D. (2024) Polypyrrole Derivatives: Preparation, Properties and Application. Polymers (Basel) 16(16):2233. https://doi.org/10.3390/polym16162233. PMID: 39204453; PMCID: PMC11360100..
[10] Karimadom BR, Kornweitz H. (2021) Mechanism of Producing Metallic Nanoparticles, with an Emphasis on Silver and Gold Nanoparticles, Using Bottom-Up Methods. Molecules 26(10):2968. https://doi.org/10.3390/molecules26102968. PMID: 34067624; PMCID: PMC8156005..
[11] Peck KA, Lien J, Su M, Stacy AD, Guo T. (2023) Bottom-Up Then Top-Down Synthesis of Gold Nanostructures Using Mesoporous Silica-Coated Gold Nanorods. ACS Omega 8(45):42667-42677. https://doi.org/10.1021/acsomega.3c05444. PMID: 38024760; PMCID: PMC10652254..
[12] Wacławska M, Dzwolak W. (2025) Shedding Light on the Proteinaceous Envelope Protecting Luminescent Gold Nanoclusters: A Review. Photochem 5(1):3. https://doi.org/10.3390/photochem5010003.
[13] Negishi Y, Takasugi Y, Sato S, Yao H, Kimura K, Tsukuda T. (2004) Magic-numbered Au(n) clusters protected by glutathione monolayers (n = 18, 21, 25, 28, 32, 39): isolation and spectroscopic characterization. J Am Chem Soc 126(21):6518-9. https://doi.org/10.1021/ja0483589. PMID: 15161256.
[14] Chu H-W, Demissie GG, Huang C-C, Anand A. (2025) Protein-templated metal nanoclusters for chemical sensing. Front Anal Sci 5:1510588. https://doi.org/10.3389/frans.2025.1510588.
[15] Bharadwaj KK, Rabha B, Pati S, Sarkar T, Choudhury BK, et al. (2021) Green Synthesis of Gold Nanoparticles Using Plant Extracts as Beneficial Prospect for Cancer Theranostics. Molecules 26(21):6389. https://doi.org/10.3390/molecules26216389. PMID: 34770796; PMCID: PMC8586976.
[16] Govindaraju S, Yun K. (2018) Synthesis of gold nanomaterials and their cancer-related biomedical applications: an update. 3 Biotech 8(2):113. https://doi.org/10.1007/s13205-018-1137-y . PMID: 29430374; PMCID: PMC5799112
[17] Paliwal R, Babu RJ, Palakurthi S. (2014) Nanomedicine scale-up technologies: feasibilities and challenges. AAPS PharmSciTech 15(6):1527-34. https://doi.org/10.1208/s12249-014-0177-9. PMID: 25047256; PMCID: PMC4245446.
[18] Yang Z, Yang X, Guo Y, Kawasaki H. (2023) A Review on Gold Nanoclusters for Cancer Phototherapy. ACS Appl Bio Mater 6(11):4504-4517. https://doi.org/10.1021/acsabm.3c00518. PMID: 37828759..
[19] Aikens CM. (2018) Electronic and Geometric Structure, Optical Properties, and Excited State Behavior in Atomically Precise Thiolate-Stabilized Noble Metal Nanoclusters. Acc Chem Res 51(12):3065-3073. https://doi.org/10.1021/acs.accounts.8b00364. PMID: 30444598..
[20] Qiu X, Xu J, Cardoso Dos Santos M, Hildebrandt N. (2022) Multiplexed Biosensing and Bioimaging Using Lanthanide-Based Time-Gated Förster Resonance Energy Transfer. Acc Chem Res 55(4):551-564. https://doi.org/10.1021/acs.accounts.1c00691. Epub 2022 Jan 27. PMID: 35084817.
[21] Wang W, Huang Y, Zhang Y, Xiao S, Luo D, et al. (2025) Research progress of multifunctional inorganic nanomaterials for tumor photothermal therapy. Nanoscale 17(38):21938-21960. https://doi.org/10.1039/D5NR02839F. PMID: 40948016.
[22] Zhou M, Zhang R, Huang M, Lu W, Song S, et al. (2010) A chelator-free multifunctional [64Cu]CuS nanoparticle platform for simultaneous micro-PET/CT imaging and photothermal ablation therapy. J Am Chem Soc 132(43):15351-8. https://doi.org/10.1021/ja106855m. PMID: 20942456; PMCID: PMC2966020.
[23] Landsman ML, Kwant G, Mook GA, Zijlstra WG. (1976) Light-absorbing properties, stability, and spectral stabilization of indocyanine green. J Appl Physiol 40(4):575-83. https://doi.org/10.1152/jappl.1976.40.4.575 . PMID: 776922.
[24] Link S, El-Sayed MA. (2000) Shape and size dependence of radiative, non-radiative and photothermal properties of gold nanocrystals. Int Rev Phys Chem 19(3):409-453. https://doi.org/10.1080/01442350050034180.
[25] Wang T, Xiao D. (2021) Rapid synthesis of fluorescent bovine serum albumin-gold nanoclusters complex for glutathione determination. Mikrochim Acta 188(6):193. https://doi.org/10.1007/s00604-021-04844-8. PMID: 34009425.
[26] You JG, Tseng WL. (2019) Peptide-induced aggregation of glutathione-capped gold nanoclusters: A new strategy for designing aggregation-induced enhanced emission probes. Anal Chim Acta 1078:101-111. https://doi.org/10.1016/j.aca.2019.05.069 . PMID: 31358207.
[27] Behzadi S, Serpooshan V, Tao W, Hamaly MA, Alkawareek MY, et al. (2017) Cellular uptake of nanoparticles: journey inside the cell. Chem Soc Rev 46(14):4218-4244. https://doi.org/10.1039/c6cs00636a . PMID: 28585944; PMCID: PMC5593313 .
[28] Chithrani BD, Chan WC. (2007) Elucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapes. Nano Lett 7(6):1542-50. https://doi.org/10.1021/nl070363y . PMID: 17465586.
[29] Matulionyte M, Dapkute D, Budenaite L, Jarockyte G, Rotomskis R. (2017) Photoluminescent Gold Nanoclusters in Cancer Cells: Cellular Uptake, Toxicity, and Generation of Reactive Oxygen Species. Int J Mol Sci 18(2):378. https://doi.org/10.3390/ijms18020378 . PMID: 28208642; PMCID: PMC5343913.
[30] Van der Sanden N, Paun RA, Yitayew MY, Boyadjian O, Tabrizian M. (2024) An investigation of the effect of the protein corona on the cellular uptake of nanoliposomes under flow conditions using quartz crystal microgravimetry with dissipation. Nanoscale Adv 7(1):169-184. https://doi.org/10.1039/d4na00783b. PMID:39569329; PMCID: PMC11575535.
[31] Dridi N, Jin Z, Perng W, Mattoussi H. (2024) Probing Protein Corona Formation around Gold Nanoparticles: Effects of Surface Coating. ACS Nano 18(12):8649-8662. https://doi.org/10.1021/acsnano.3c08005 . PMID: 38471029.
[32] Mosquera J, García I, Henriksen-Lacey M, Martínez-Calvo M, Dhanjani M, et al. (2020) Reversible Control of Protein Corona Formation on Gold Nanoparticles Using Host-Guest Interactions. ACS Nano 14(5):5382-5391. https://doi.org/10.1021/acsnano.9b08752 . PMID: 32105057; PMCID: PMC7254833.
[33] Landsiedel R, Fabian E, Ma-Hock L, van Ravenzwaay B, Wohlleben W, et al. (2012) Toxico-/biokinetics of nanomaterials. Arch Toxicol 86(7):1021-60. https://doi.org/10.1007/s00204-012-0858-7 . PMID: 22576463.
[34] Tee JK, Peng F, Ho HK. (2019) Effects of inorganic nanoparticles on liver fibrosis: Optimizing a double-edged sword for therapeutics. Biochem Pharmacol 160:24-33. https://doi.org/10.1016/j.bcp.2018.12.003 . PMID: 30529191.
[35] Xu N, Zhang X, Qi T, Wu Y, Xie X, et al. (2022). Biomedical applications and prospects of temperature‐orchestrated photothermal therapy. MedComm–Biomaterials and Applications, 1(2), e25. https://doi.org/10.1002/mba2.25.
[36] Wang Z, Hasan I, Zhang Y, Peng T, Guo B. (2025) Photothermal Combination Therapy for Metastatic Breast Cancer: A New Strategy and Future Perspectives. Biomedicines 13(10):2558. https://doi.org/10.3390/biomedicines13102558 . PMID: 41153838; PMCID: PMC12561961 .
[37] Baghdasaryan A, Dai H. (2025) Molecular Gold Nanoclusters for Advanced NIR-II Bioimaging and Therapy. Chem Rev 125(11):5195-5227. https://doi.org/10.1021/acs.chemrev.4c00835 . PMID: 40435324; PMCID: PMC12164276.
[38] Prajapati S, Yadav S, Khan J. (2024) Theranostic and Combined Approaches Exploiting Multifunctional Gold Nanoclusters in Tumoral Ecosystems: A Paradigm Shift in Precision Oncology. Curr Radiopharm 17(4):330-340. https://doi.org/10.2174/0118744710283369240328082442. PMID: 38571350.
[39] Xiao G, Zhao Y, Wang X, Zeng C, Luo F, Jing J. (2023) Photothermally sensitive gold nanocage augments the antitumor efficiency of immune checkpoint blockade in immune "cold" tumors. Front Immunol 14:1279221. https://doi.org/10.3389/fimmu.2023.1279221. PMID: 37942337; PMCID: PMC10628457.
[40] Peng J, Qi T, Liao J, Chu B, Yang Q, et al. (2014) Mesoporous magnetic gold "nanoclusters" as theranostic carrier for chemo-photothermal co-therapy of breast cancer. Theranostics 4(7):678-92. https://doi.org/10.7150/thno.7869. PMID: 24883118; PMCID: PMC4038750.
[41] He Z, Wang Q, Zhang N, Yan J, Li L, et al. (2022) Gold nanorods/tetrahedral DNA composites for chemo-photothermal therapy. Regen Biomater 9:rbac032. https://doi.org/10.1093/rb/rbac032 . PMID: 35668924; PMCID: PMC9163824.
[42] de la Fuente-Jiménez JL, Rodríguez-Rivas CI, Mitre-Aguilar IB, Torres-Copado A, García-López EA, et al. (2023) A Comparative and Critical Analysis for In Vitro Cytotoxic Evaluation of Magneto-Crystalline Zinc Ferrite Nanoparticles Using MTT, Crystal Violet, LDH, and Apoptosis Assay. Int J Mol Sci 24(16):12860. https://doi.org/10.3390/ijms241612860 . PMID: 37629040; PMCID: PMC10454666.
[43] Barriault C, Audet M, Yousef IM, Tuchweber B. (1995) Effect of agents which modify reticuloendothelial system function on acute phalloidin-induced lethality and hepatotoxicity in mice. Toxicol Appl Pharmacol 131(2):206-15. https://doi.org/10.1006/taap.1995.1063 . PMID: 7716763.
[44] Zhou C, Long M, Qin Y, Sun X, Zheng J. (2011) Luminescent gold nanoparticles with efficient renal clearance. Angew Chem Int Ed Engl 50(14):3168-72. https://doi.org/10.1002/anie.201007321 . PMID: 21374769; PMCID: PMC3095852 .
[45] Liu J, Yu M, Zhou C, Yang S, Ning X, Zheng J. (2013) Passive tumor targeting of renal-clearable luminescent gold nanoparticles: long tumor retention and fast normal tissue clearance. J Am Chem Soc 135(13):4978-81. https://doi.org/10.1021/ja401612x . PMID: 23506476; PMCID: PMC4127425.
[46] Du B, Jiang X, Das A, Zhou Q, Yu M, et al. (2017) Glomerular barrier behaves as an atomically precise bandpass filter in a sub-nanometre regime. Nat Nanotechnol 12(11):1096-1102. https://doi.org/10.1038/nnano.2017.170 . PMID: 28892099; PMCID: PMC5679252.
[47] Yu Y, Yang T, Sun T. (2020) New insights into the synthesis, toxicity and applications of gold nanoparticles in CT imaging and treatment of cancer. Nanomedicine (Lond) 15(11):1127-1145. https://doi.org/10.2217/nnm-2019-0395 . PMID: 32329396.
[48] European Medicines Agency (EMA). Joint Strategy of the European Medicines Regulatory Network to 2028: Seizing Opportunities in a Changing Medicines Landscape. EMA/846118/2020 (Updated 2025/2026). Available from: ema.europa.eu
[49] Mangla B, Kumar P, Javed S, Pathan T, Ahsan W, Aggarwal G. (2025) Regulating nanomedicines: challenges, opportunities, and the path forward. Nanomedicine (Lond) 20(15):1911-1927. https://doi.org/10.1080/17435889.2025.2533107 . PMID: 40657903.
[50] Jiang X, Du B, Huang Y, Yu M, Zheng J. (2020) Cancer Photothermal Therapy with ICG-Conjugated Gold Nanoclusters. Bioconjug Chem 31(5):1522-1528. https://doi.org/10.1021/acs.bioconjchem.0c00172 . PMID: 32353229; PMCID: PMC8667163.
[51] Huang H, du Toit H, Panariello L, Mazzei L, Gavriilidis A. (2021) Continuous synthesis of gold nanoparticles in micro-and millifluidic systems. Phys Sci Rev 6(3):20170119. https://doi.org/10.1515/psr-2017-0119
[52] Li J, Feng Y, Ge J. (2025) Recent Advancements in Bionic Olfactory Biosensors: Components, Applications, and Future Perspectives. Chem Bio Eng 2(10):576-592. https://doi.org/10.1021/cbe.5c00042 . PMID: 41158608; PMCID: PMC12557451.
[53] Zhang Q, Hou D, Wen X, Xin M, Li Z, et al. (2022) Gold nanomaterials for oral cancer diagnosis and therapy: Advances, challenges, and prospects. Mater Today Bio 15:100333. https://doi.org/10.1016/j.mtbio.2022.100333 . PMID: 35774196; PMCID: PMC9237953.
[54] Mehta M, Bui TA, Yang X, Aksoy Y, Goldys EM, Deng W. (2023) Lipid-Based Nanoparticles for Drug/Gene Delivery: An Overview of the Production Techniques and Difficulties Encountered in Their Industrial Development. ACS Mater Au 3(6):600-619. https://doi.org/10.1021/acsmaterialsau.3c00032 . PMID: 38089666; PMCID: PMC10636777.
[55] Chou WC, Canchola A, Zhang F, Lin Z. (2025) Machine Learning and Artificial Intelligence in Nanomedicine. Wiley Interdiscip Rev Nanomed Nanobiotechnol 17(4):e70027. https://doi.org/10.1002/wnan.70027 . PMID: 40813104; PMCID: PMC12353477.
[56] Zaki M, Prinz C, Ruehle B. (2025) A Self-Driving Lab for Nano- and Advanced Materials Synthesis. ACS Nano 19(9):9029-9041. https://doi.org/10.1021/acsnano.4c17504 . PMID: 39995288; PMCID: PMC11912568.
[57] Meng Y, Sui L, Xu T, Zhao H, Yuan Q, Sun L. (2025) Research and Application Prospect of Nanomedicine in Kidney Disease: A Bibliometric Analysis From 2003 to 2024. Int J Nanomedicine 20:3007-3030. https://doi.org/10.2147/IJN.S510016. PMID: 40093546; PMCID: PMC11910916.
[58] Chen Y, Tang D. (2025) Precision nanomaterials in colorectal cancer: advancing photodynamic and photothermal therapy. RSC Adv 15(33):26583-26600. https://doi.org/10.1039/d5ra03996g . PMID: 40727285; PMCID: PMC12291162.
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
All articles in the World Journal of Experimental Biosciences are published under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0) ( (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.