Controlling Imipenem-Resistant Pseudomonas aeruginosa in Hospitals: Effect of Improved Infection Control Measures from an Interrupted Time-Series Study
Control of Imipenem-Resistant Pseudomonas aeruginosa in Hospitals
DOI:
https://doi.org/10.65329/wjeb.v14.01.03Keywords:
Active surveillance; Antimicrobial resistance, Hospital-acquired infection; Infection prevention and control; Imipenem-resistant Pseudomonas aeruginosaAbstract
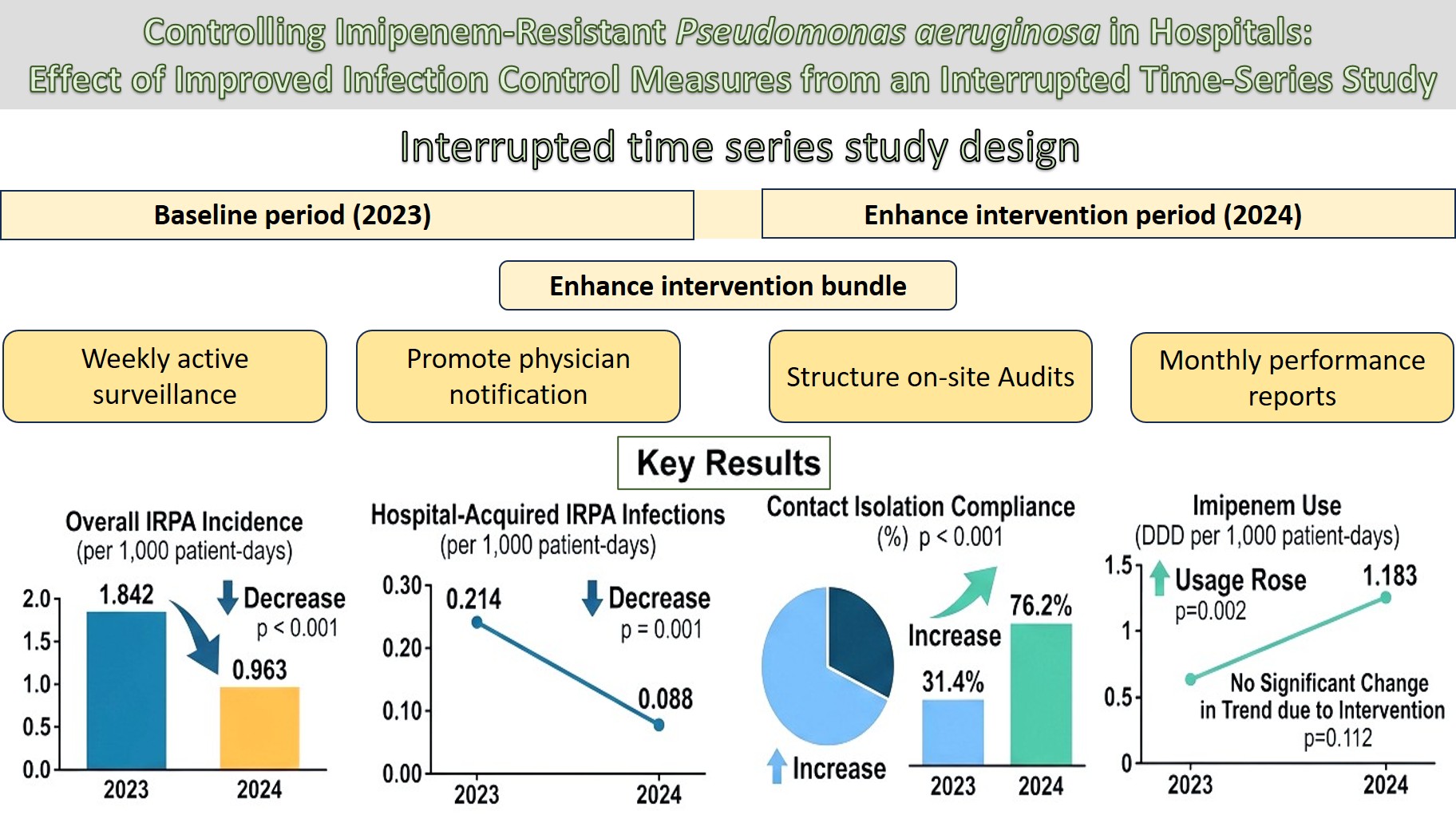
Imipenem-resistant Pseudomonas aeruginosa (IRPA) causes serious healthcare-associated infections and negatively affects clinical outcomes. This study aims to determine if a structured bundle of enhanced infection-control measures can effectively reduce hospital-acquired IRPA cases at a tertiary care hospital. A retrospective interrupted time series analysis was conducted over two consecutive twelve-month periods: (1) 2023 served as the baseline period; (2) 2024 as the enhanced intervention period. The interventions included weekly active surveillance, prompt notification to physicians after positive culture results, structured on-site audits with a standardized checklist for isolating multidrug-resistant organisms, and monthly departmental performance reports. A total of 850 clinical isolates were analyzed. The main outcomes measured were the overall IRPA incidence, hospital-acquired IRPA infection rates, adherence to contact precautions for IRPA patients, and total Imipenem use, each expressed as a rate per 1,000 patient-days. Segmented linear regression was used for the interrupted time series analysis. IRPA incidence decreased from 1.842 to 0.963 cases per 1,000 patient-days (p < 0.001), and hospital-acquired IRPA infection rates dropped from 0.214 to 0.088 per 1,000 patient-days (p = 0.001). Contact isolation compliance increased from 31.4% to 76.2% (p < 0.001). Imipenem use rose to 1.183 defined daily doses per 1,000 patient-days per quarter (p = 0.002), with no significant change in this trend due to the intervention (p = 0.112). The implementation of a structured infection control bundle focusing on weekly surveillance and real-time notification led to a significant clinical and statistical reduction in hospital-acquired IRPA, despite increased Imipenem use. These findings offer practical evidence for resource-limited tertiary care settings.
References
[1] Forrester JD, Maggio PM, Tennakoon L. (2022) Cost of Health Care-Associated Infections in the United States. J Patient Saf 18(2):e477-e479. doi: https://doi.org/10.1097/PTS.0000000000000845. PMID: 33881808.
[2] Ghali H, Saad OKB, Bhiri S, Balhi S, Azouzi F, et al. (2025) Epidemiology and risk factors of healthcare-associated urinary tract infections: a prospective study in a Tunisian tertiary hospital. Sci Rep 15(1):29948. doi: https://doi.org/10.1038/s41598-025-03971-z. PMID: 40817091; PMCID: PMC12356833.
[3] Petrakis V, Rafailidis P, Panopoulou M, Konstantinidis T, Babaka N, et al. (2025) Efficacy of Imipenem/Cilastatin/Relebactam in infections caused by Difficult-to-Treat Gram-Negative Bacteria: A Retrospective Analysis of Real-world data in Greece. Infez Med 33(4):422-434. doi: https://doi.org/10.53854/liim-3304-7 . PMID: 41384153; PMCID: PMC12695162.
[4] Garvey MI, Wilkinson MAC, Holden KL, Martin T, Parkes J, Holden E. (2019) Tap out: reducing waterborne Pseudomonas aeruginosa transmission in an intensive care unit. J Hosp Infect 102(1):75-81. doi: https://doi.org/10.1016/j.jhin.2018.07.039. PMID: 30071267.
[5] Flores-Vega VR, Partida-Sanchez S, Ares MA, Ortiz-Navarrete V, Rosales-Reyes R. (2024) High-risk Pseudomonas aeruginosa clones harboring β-lactamases: 2024 update. Heliyon 11(1):e41540. doi: https://doi.org/10.1016/j.heliyon.2024.e41540. PMID: 39850428; PMCID: PMC11754179.
[6] Bonomo RA, Perez F, Hujer AM, Hujer KM, Vila AJ. (2024) The Real Crisis in Antimicrobial Resistance: Failure to Anticipate and Respond. Clin Infect Dis 78(6):1429-1433. doi: https://doi.org/10.1093/cid/ciad758. PMID: 38289748.
[7] Zurita J, Sevillano G, Solís MB, Paz Y Miño A, Alves BR, et al. (2024) Pseudomonas aeruginosa epidemic high-risk clones and their association with multidrug-resistant. J Glob Antimicrob Resist 38:332-338. doi: https://doi.org/10.1016/j.jgar.2024.07.003. PMID: 39019398.
[8] Sati H, Carrara E, Savoldi A, Hansen P, Garlasco J, et al. (2025) WHO Bacterial Priority Pathogens List Advisory Group. The WHO Bacterial Priority Pathogens List 2024: a prioritisation study to guide research, development, and public health strategies against antimicrobial resistance. Lancet Infect Dis 25(9):1033-1043. doi: https://doi.org/10.1016/S1473-3099(25)00118-5. PMID: 40245910; PMCID: PMC12367593.
[9] GBD 2021 Antimicrobial Resistance Collaborators (2024). Global burden of bacterial antimicrobial resistance 1990-2021: a systematic analysis with forecasts to 2050. Lancet (London, England) 404(10459), 1199–1226. doi: https://doi.org/10.1016/S0140-6736(24)01867-1.
[10] Magiorakos AP, Burns K, Rodríguez Baño J, Borg M, Daikos G, et al. (2017) Infection prevention and control measures and tools for the prevention of entry of carbapenem-resistant Enterobacteriaceae into healthcare settings: guidance from the European Centre for Disease Prevention and Control. Antimicrob Resist Infect Control 6:113. doi: https://doi.org/10.1186/s13756-017-0259-z. PMID: 29163939; PMCID: PMC5686856.
[11] Assiri MA, Alqarni HE, Asiri ZA, Asiri AM, Alasmari AAM, et al. (2024) Infection Control in Clinical Nursing: Best Practices for Reducing Nosocomial Infections. The Review of Diabetic Studies 51-63. doi: https://doi.org/10.70082/p4vyqf32 .
[12] Kousouli E, Zarkotou O, Politi L, Polimeri K, Vrioni G, et al. (2018) Infection control interventions affected by resource shortages: impact on the incidence of bacteremias caused by carbapenem-resistant pathogens. Eur J Clin Microbiol Infect Dis 37(1):43-50. doi: https://doi.org/10.1007/s10096-017-3098-1. PMID: 28879405.
[13] CLSI. (2023) Performance Standards for Antimicrobial Susceptibility Testing. 33rd ed. CLSI supplement M100. Clinical and Laboratory Standards Institute.
[14] Ahmadi MH, Fagheei Aghmiyuni Z, Bakhti S. (2025) Worldwide threatening prevalence of carbapenem-resistant Pseudomonas aeruginosa. Epidemiol Infect 153:e114. doi: https://doi.org/10.1017/S0950268825100332. PMID: 40970428; PMCID: PMC12529429.
[15] Tamma PD, Heil EL, Justo JA, Mathers AJ, Satlin MJ, Bonomo RA. (2024) Infectious Diseases Society of America 2024 Guidance on the Treatment of Antimicrobial-Resistant Gram-Negative Infections. Clin Infect Dis 7:ciae403. doi: https://doi.org/10.1093/cid/ciae403. PMID: 39108079.
[16] García-Fernández S, Calvo J, Cercenado E, Suárez-Barrenechea AI, Fernández-Billón M, (2023) Activity of imipenem/relebactam against Enterobacterales and Pseudomonas aeruginosa in Spain. SMART 2016-2020. Rev Esp Quimioter 36(3):302-309. doi: https://doi.org/10.37201/req/007.2023, PMID: 36951688, PMCID: PMC10238800
[17] Wong SC, Chau PH, So SY, Lam GK, Chan VW, et al. (2022) Control of Healthcare-Associated Carbapenem-Resistant Acinetobacter baumannii by Enhancement of Infection Control Measures. Antibiotics (Basel) 11(8):1076. doi: https://doi.org/10.3390/antibiotics11081076. PMID: 36009945; PMCID: PMC9405119.
[18] Cho OH, Bak MH, Baek EH, Park KH, Kim S, Bae IG. (2014) Successful control of carbapenem-resistant Acinetobacter baumannii in a Korean university hospital: a 6-year perspective. Am J Infect Control 42(9):976-9. doi: https://doi.org/10.1016/j.ajic.2014.05.027. PMID: 25179329.
[19] Martínez DA, Cai J, Lin G, Goodman KE, Paul R, et al. (2023) MInD-Healthcare. Modelling interventions and contact networks to reduce the spread of carbapenem-resistant organisms between individuals in the ICU. J Hosp Infect 136:1-7. doi: https://doi.org/10.1016/j.jhin.2023.02.016 . PMID: 36907332; PMCID: PMC10315994.
[20] Ge T, Feng H, Ni L, Wang F, Zhan Q, et al. (2025) Optimization of environmental surveillance-driven carbapenem-resistant organisms infection prevention and control strategies in intensive care units: A 4-year multicampus prospective intervention study. Am J Infect Control 21:S0196-6553(25)00726-6. doi: https://doi.org/10.1016/j.ajic.2025.11.014 .11.014. Epub ahead of print. PMID: 41276087.
[21] Tamma PD, Heil EL, Justo JA, Mathers AJ, Satlin MJ, Bonomo RA. (2024) Infectious Diseases Society of America 2024 Guidance on the Treatment of Antimicrobial-Resistant Gram-Negative Infections. Clin Infect Dis 7:ciae403. doi: https://doi.org/10.1093/cid/ciae403 . PMID: 39108079.
[22] Büchler AC, Klaassen CHW, de Goeij I, Vos MC, Voor In 't Holt AF, Severin JA. (2025) Outbreak investigations of contact patients and the hospital environment after detection of carbapenemase-producing Pseudomonas aeruginosa on general hospital wards. J Hosp Infect 159:11-19. doi: https://doi.org/10.1016/j.jhin.2025.02.009. PMID: 39986654.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Authors

This work is licensed under a Creative Commons Attribution 4.0 International License.
All articles in the World Journal of Experimental Biosciences are published under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0) ( (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.